Substances dont change temperature when they change states. The boiling point of water depends on the atmospheric pressure which changes according to elevation.
/BoilingWater-58dd1c2a5f9b5846837d2a23.jpg)
What Is The Boiling Point Of Water
Heating a substance in the solid state will cause it to melt which changes it to the liquid state.

How does the temperature of water change as it boils. Continued heating will cause the substance to evaporate or boil which changes it to the gas. Water for example would stay liquid and increase in temperature until it started boiling then it would stop increasing until it turned. It takes longer to boil a large beaker of water than a small beaker.
Since this is a naturally-occurring condition in high altitude regions the boiling point of water is reduced and thus it attains the boiling temperature quicker than at the ground level with the same heat. Simply put it boils faster. Therefore the temperature of the liquid remains constant during boiling.
You can crank the heat as high as you like. Time for water changing from a liquid to a gas called a heating curve shows a constant temperature as long as water is boiling. Water boils at sea level at an ambient pressure of 1 bar at 100 C.
This breaking of bonds between water molecules consumes any additional thermal energy added so that water at boiling temperature does not increase in temperature until the phase change is complete. As we move higher into the atmosphere and the atmospheric pressure drops so too does the amount of vapour pressure required for a liquid to boil. With that much less pressure you dont need to apply as much heat to push vapor pressure beyond the surrounding atmospheric pressure in other words water boils at a lower temperature.
In Denver the atmospheric pressure is only about 12 pounds per square inch compared with 147 pounds per square inch in Los Angeles. Boiling means it is all changing into a gas. At sea level water boils at 100 c 212 f and freezes at 0 c 32 f.
At that point the energy. This is an example of boiling point elevation and it is not exclusive to water. If you heat water its temperature will increase until it reaches100 degrees C at which point it starts to boil.
This shows the phenomenon that water begins to boil at significantly lower temperatures than one is used to at lower altitudes. The temperature needed to boil will increase about 05 C for every 58 grams of dissolved salt per kilogram of water. If you place pressure on the water.
A lower boiling point means that food. The same is true of water. At sea level vapour pressure is equal to the atmospheric pressure at 100 C and so this is the temperature at which water boils.
The water starting out at roughly 212 F 100 C instantly turns to snow upon hitting the air and blows away. Click to see full answer. The water may boil more vigorously and convert into steam more quickly but it wont get hotter.
Energy added to liquid water will raise its temperature to 100 C. The temperature at which water boils becomes lower where there is lower air pressure so water would boil at a lower temperature near the top of a high mountain. Boiling means itis all changing into a gas.
At sea level at a pressure of 1013 bar water begins to boil at a temperature of 100 C. Water boils at a lower temperature as you gain altitude eg going higher on a mountain and boils at a higher temperature if you increase atmospheric pressure coming back down to sea level or going below it. As the altitude increases the atmospheric pressure pushing down on water decreases which allows the water to boil at lower temperatures.
More heat just makes it boils faster. To boil water we must increase its temperature to 100C. Water boils when the thermal energy in the water which is a type of kinetic energy which causes the water molecules to move around exceeds the strength of the hydrogen bonds between the molecules causing them to separate from the other molecules.
A graph of temperature vs. Similarly water molecules have an easy time escaping off the surface when the air pressure above them is less. This is because the large beaker contains more water and needs to gain.
At a lower pressure like in the mountains the boiling point is a lower temperature. But whatever the boiling point is when water reaches it and undergoes a phase transition into water vapor steam the temperature stops rising. When water boils its temperature stays constant.
It occurs any time you add a nonvolatile solute such as salt to a solvent such as water. Furthermore how cold does it have to be for boiling water to freeze in air. For example water will remain at 100ºC at a pressure of 1 atm or 1013 kPa while boiling.
When it reaches the boiling point it stays the same temperature because energy is being released and regulated by letting off steam. One of the most significant changes that occur in high altitude areas concerning cooking is the boiling point of water.
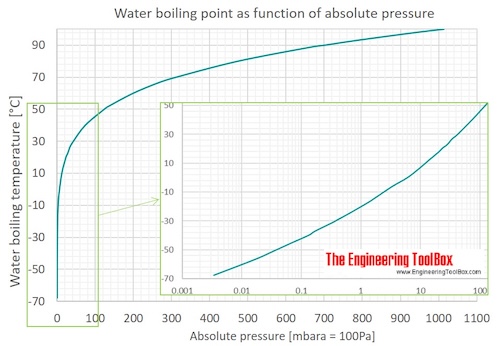
Water Boiling Points At Vacuum Pressure
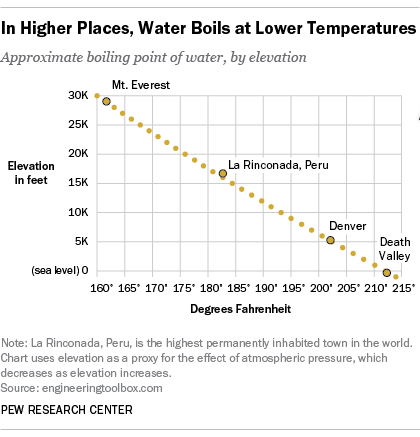
Does Water S Boiling Point Change With Altitude Americans Aren T Sure Pew Research Center
