This was the first observation of the photoelectric effect. Einstein proposed photons to be quanta of EM radiation having energy E hf where f is the frequency of the radiation.
O Light is absorbed permanently by electrons in a metal The energy of light is quantized by its frequency O Light behaves as a wave in this experiment.

What did the photoelectric experiment demonstrate. Thompson found that the amount of current varied with the intensity and frequency of the radiation used. For example as shown below a typical experiment in science demonstrations is to shine ultraviolet light on a zinc metal plate and observe that electrons are emitted from the zinc. But both the observations of the photoelectric effect could not be explained by Maxwells electromagnetic wave theory of light.
A year later Wilhelm Hallwachs confirmed these results and showed that UV light on a Zinc plate connected to a battery generated a current because of electron emission. The notion of light quantization was first introduced by Planck. A quantitative measure of the photoelectric effect came in.
So if light is classical wave predict that just puts energy into plate heats up get diode current voltage curve. At the high photon energies comparable to the electron rest energy of 511 keV Compton scattering may occur pair production may take place at energies over 1022 MeV. It is a vacuum sealed tube with a cathode and anode on one side.
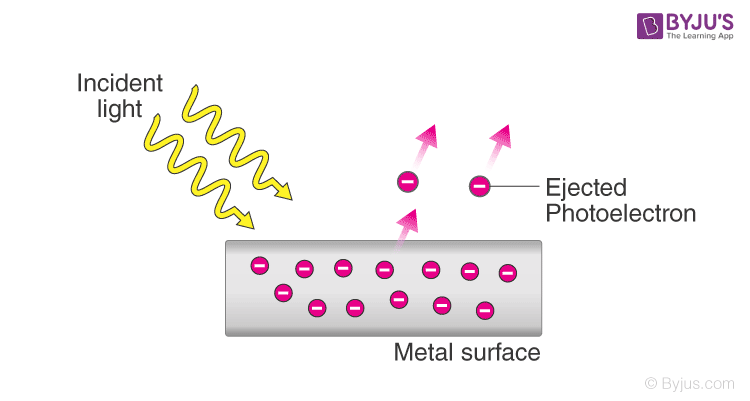
The photoelectric effect is the emission of electrons when electromagnetic radiation such as light hits a materialElectrons emitted in this manner are called photoelectrons. Of determining the ratio of Plancks constant h against the electron charge e ie. Kevin Mcgill Answer The photoelectric effect refers to the emission or ejection of electrons from the surface of generally a metal in response to incident light.
An experiment to demonstrate that water is formed by a reaction between oxygen and hydrogen 24 June 1783. The basic physical process underlying this effect is the emission of electrons in metals exposed to light. He also proved these waves were polarised.
Thomsons cathode ray experiment was a set of three experiments that assisted in discovering electrons. What is the Photoelectric Effect. The photoelectric effect can be demonstrated using a gold leaf electroscope The photoelectric effect is where electrons are emitted from certain materials when light ie.
0 Voltage Also takes time to heat up. This successful experiment was followed up by a series of others through which Hertz demonstrated that these electromagnetic waves could be reflected from a metal mirror and refracted as they passed through a prism made from pitch thus proving that these waves behaved similarly to light waves. Energy contained within the incident light is absorbed by electrons within the metal giving the electrons sufficient energy to be knocked out of that is emitted from the surface of.
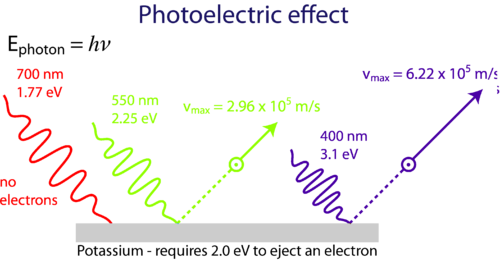
Then in 1899 in England JJ. The photoelectric effect At the turn of the century physicists who experimented with electricity had noticed something about the interaction of light metals and electric current. The photoelectric effect occurs with photons having energies from a few electronvolts to over 1 MeV.
This relation between light and electricity hence photoelectric was clarified in 1902 by another German physicist Philipp Lenard. The concept of the photoelectric effect was first documented in 1887 by Heinrich Hertz and later by Lenard in 1902. He demonstrated that electrically charged particles are liberated from a metal surface when it is illuminated and that these particles are identical to electrons which had been discovered by the British physicist Joseph John Thomson in 1897.
First demonstrated successfully by Milikan2 the photoelectric experiment has become a staple in advanced undergraduate physics labs using more advanced equipment with the primary goal 1. The phenomenon is studied in condensed matter physics and solid state and quantum chemistry to draw inferences about the properties of atoms molecules and solids. Its validity is based on solid experimental evidence most notably the photoelectric effect.
The effect has found use in electronic devices. The photoelectric effect was first observed in the 1880s At that time scientists thought light was a wave and this explaine many properties of light eg. The photoelectric effect is a phenomenon where electrons are emitted from the metal surface when the light of sufficient frequency is incident upon.
The photoelectric effect is the process in which EM radiation ejects electrons from a material. He did this using a cathode ray tube or CRT. Reflection refraction and defraction.
Photoelectric effect experiment apparatus. Thompson demonstrated that ultraviolet light hitting a metal surface caused the ejection of electrons. The photoelectric effect is an effect where electrons are emitted from certain materials when light of a certain minimum wavelength arrives at the material.
QUESTION 1 What did the Photoelectric Effect demonstrate.
Solved 30 Which Type Of Experiment Demonstrates That Lig Chegg Com

Photoelectric Effect Learning Outcomes Describe The Photoelectric Effect Demonstrate The Photoelectric Effect Discuss The Nature Of The Photon Ppt Download

Photoelectric Effect Wikipedia
Photoelectric Effect Chemistry For Non Majors
The Photoelectric Effect Detailed Explanation With Illustrations

