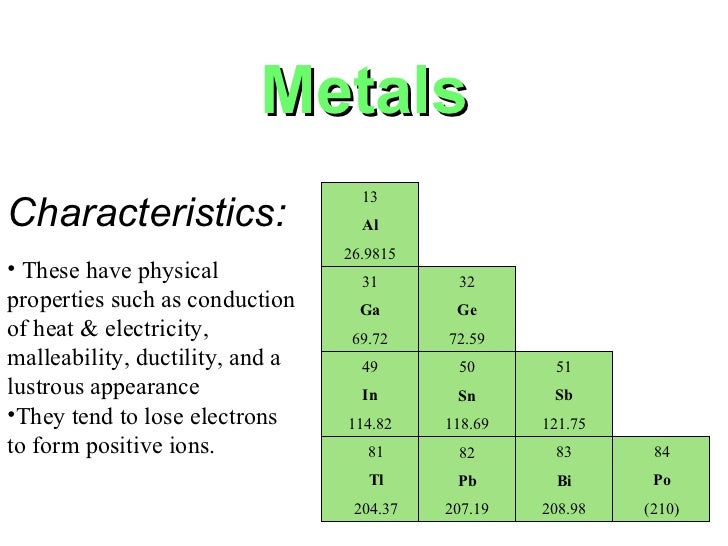
Most of them have low-density. Metals shiny solids are room temperature except mercury which is a shiny liquid element with characteristic high melting points and densities.
Definition Of Metals Chemistry Dictionary
The elements in group 1 are called the alkali metals.
Characteristics of metals on the periodic table. List and explain the properties of metals Remember that metals are on the left and bottom of the periodic table. Metals prefer to eliminate electrons in their last shell. Good conductors of heat.
Some elements have properties that are not typical. Most elements are metalsThey are usually shiny very dense and only melt at high temperatures. The highest density alkali metal is Cesium Cs element.
The reactive nonmetals near the metalloids show some incipient metallic character such as the metallic appearance of graphite black phosphorus selenium and iodine. The lowest density of alkali metal is lithium Li element. Transition metals Because this group contains so many elements they exhibit a wide range of properties.
Group 1 - physical properties Group 1 contains elements placed in a vertical column on the far left of the periodic table. Solid at room temperature with the exception of mercury usually shiny high melting point good conductor of heat good conductor of electricity low ionization energies low electronegativities malleable able to be pounded into sheets ductile. The periodic table on the left separates elements into three groups.
The periodic table of metals and nonmetals can be broken down to give you a sense of each elements characteristics. From left to right in the periodic table the nonmetals can be divided into the reactive nonmetals and the noble gases. They are malleable.
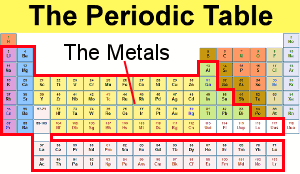
2 3 In actual practice the f-block lanthanide and actinide series are also considered transition metals and are called inner transition metals. Also many periodic tables have a stair-step line on the table identifying the element groups. The metals green in the table nonmetals orange and metalloids blue.
Most elements are metals. On the periodic table metals are separated from nonmetals by a zig-zag line stepping through carbon phosphorus selenium iodine and radon. Metals nonmetals and metalloids make up the periodic table with metals constituting the large majority of all metals.
Common characteristics of metals. Based on this group. Group 1 is on the.
S block p block d block f block. Metals Characteristics Variation in Periodic Table Metals in periodic table. The block of transitional metals which is present in middle part of the modern periodic table includes elements of group IB to VIIIB ie.
Thus metals are electropositive elements with relatively low ionization energies. The elements here are beryllium magnesium calcium strontium barium and radium. Based on the periodic trends in the last 3 sections this means that they are usually bigger more likely to lose electrons and less likely to gain electrons than the non-metals.
The Periodic Table contains a lot of useful information on the elements. The metals share several common properties including. Many of the properties of metals including a large atomic radius low ionization energy and low electronegativity are because the electrons in the valence shell of metal atoms can be removed easily.
Their shape can be easily changed into thin wires or sheets without breaking. Poor conductors of heat. Therefore they form cations.
These elements and those to the right of them are nonmetals. These metals have properties that you normally associate with the metals you encounter in everyday life. Alkaline earth metals are the elements present in main group 2 2A of the periodic table.
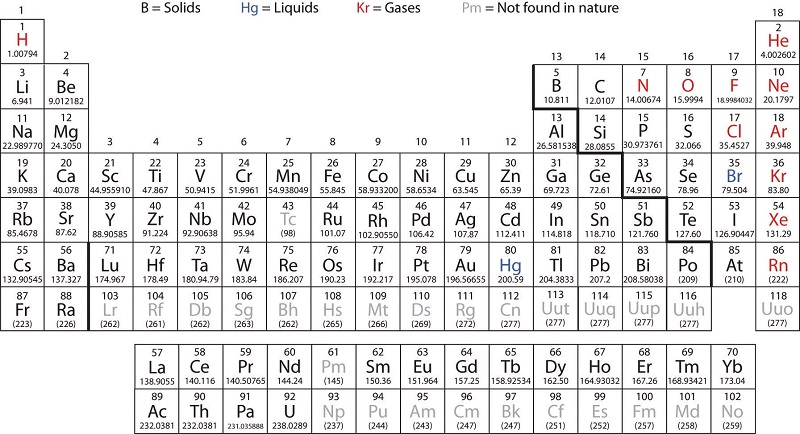
The noble gases are almost completely inert. Most often they are good conductors of heat and electricity. Lithium Li Sodium Na Potassium K because their densities are smaller than the density of water 1 gmcm³.
Many scientists describe a transition metal as any element in the d-block of the periodic table which includes groups 3 to 12 on the periodic table. This includes the alkali metals alkaline earth metals transition metals lanthanides and actinides. With the exception of hydrogen all elements that form positive ions by losing electrons during chemical reactions are called metals.
For example excluding hydrogen all of the elements in Group 1 on the very left-hand side of the periodic table are called alkali metals. They are characterized by bright luster hardness ability to resonate sound and are excellent conductors of heat and electricity. The line begins at boron B and extends down to polonium Po.
Mercury a metal has. Metals are located in all blocks in periodic table. Alkali elements that float on the water surface.
They are ductile they can be drawn into thin wires. The metalloids separate the metals and nonmetals on a periodic table. They are more dense and melt faster than metals in groups 1 and 2.
They are solid with the exception of mercury Hg a liquid. 3 to 12 these elements have last electron in d-subshell their properties are midway between those of s-block p-block so they are called Transitional elements. They are shiny good conductors of electricity and heat.
Elements of the periodic table are grouped as metals metalloids or semimetals and nonmetals. Alkali metals are soft and silvery and react violently.

Periodic Table Families Properties Uses Schoolworkhelper
19 1 Properties Of Transition Metals And Their Compounds Chemistry Libretexts

Alkali Metal Definition Properties Facts Britannica