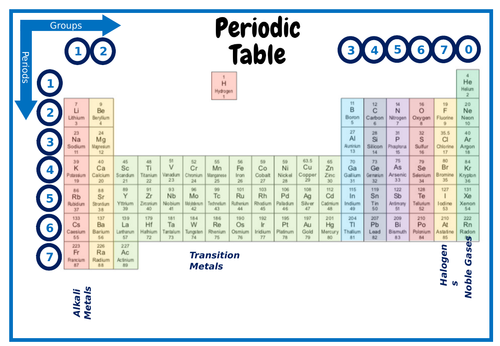
Youll find more specific groups like transition metals rare earths alkali metals alkaline earth halogens and noble gasses. Periodic table in chemistry the organized array of all the chemical elements in order of increasing atomic number.
Rows on the periodic table are referred to as periods while the columns on the periodic table are referred to as groups.
Periodic table with periods and groups labeled. Thats great to hear. The vertical columns of elements are called groups or families. The elements in a group have similar physical or chemical characteristics of the outermost electron shells of their atoms ie the same core charge because most chemical properties are dominated by the orbital location of the outermost electron.
Click on the periodic table then click on sulfur. Poor electrical and thermal conductors. Find out in this video from the Properti.
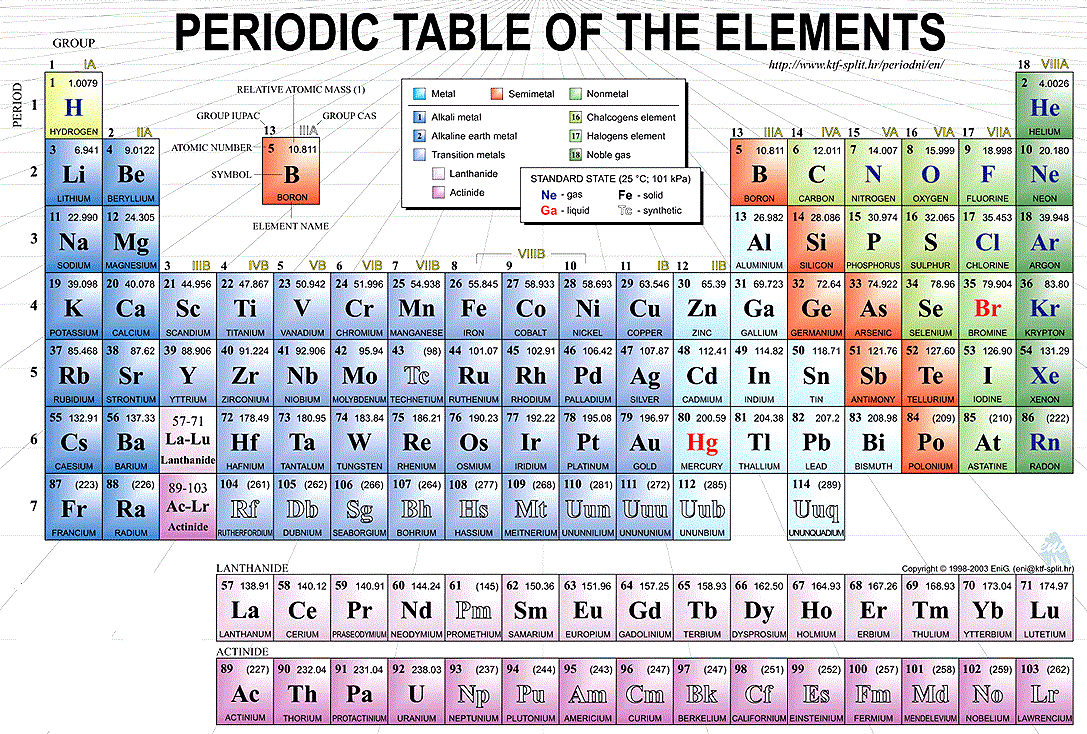
In chemistry a group also known as a family is a column of elements in the periodic table of the chemical elementsThere are 18 numbered groups in the periodic table. Masuzi October 24 2018 Uncategorized Leave a comment 12 Views. Periodic Table Labeled Groups In the late nineteenth century Russian scientific expert Dmitri Mendeleev distributed his first endeavor at gathering concoction elements as per their atomic loads.
Periods are horizontal rows across the periodic table while groups are vertical columns down the table. A new period begins when a new principal energy level begins filling with electrons. In general the periodicity of the periodic table in terms of periodic table blocks is clearly due.
Masuzi October 24 2018 Uncategorized Leave a comment 10 Views. All the different elements are arranged in a chart called the periodic table. Groups families periods and valence 3 1 periodic table mun ib reading the periodic table how to identify groups periods and.
Interactive periodic table showing names electrons and oxidation states. The elements in a group have similar physical or chemical characteristics of the outermost electron shells of their atoms ie the same core charge because. The horizontal rows of the periodic table are referred as periods while the vertical columns are referred as groups.
Period 1 has only two elements hydrogen and helium while periods 2 and 3 have 8 elements. Cir Room 9 Groups Families Periods And Valence Of The Periodic Table. Atomic Structure Web Quest Name_____ _ Period____ __ Go to 1.
Periodic Table Groups And Periods Labeled. There were just around 60 elements known at that point however Mendeleev understood that when the elements were composed by weight particular sorts of elements happened in normal spans or periods. The form of the periodic table is closely related to the electron configuration of the atoms of the elements.
Visualize trends 3D orbitals isotopes and mix compounds. Atomic number increases as you move down a group or across a period. Periods 4 and 5 have 18 elements.
Whats the difference between periods and groups in the Periodic Table and why are the elements structured this way. Groups and Periods are the columns and rows of the periodic table. Here are the main features of the table.
There are multiple ways of grouping the elements but they are commonly divided into metals semimetals metalloids and nonmetals. There are seven periods in the periodic table with each one beginning at the far left. In this section you will explore how the periodic table was put together and the two main arrangement of the periodic table focusing on the groups and periods and metals and non metals.
Updated August 10 2019 Groups and periods are two ways of categorizing elements in the periodic table. The f-block columns between groups 2 and 3 are not numbered. In the periodic table of elements there are seven horizontal rows of elements called periods.
Inner Transition Metal Simple English Wikipedia The Free. Interactive periodic table with element scarcity sri discovery dates melting and boiling points group block and period information. 29 Printable Periodic Tables Free Download ᐅ Template Lab.
Periods and groups are two important characteristics of the periodic table. Modern Periodic Table Periods And Groups Chemistry For Non Majors. An elements period number represents the highest energy level that an electron in that element possesses.
With chlorine with the help of electronic structure. The horizontal rows are called periods the vertical columns are called groups. A table in which all the known.
New Periodic Table Of Elements Labeled Groups. Scroll down to the picture of the sulfur atom and its rings. Explain what the rings show and describe sulfurs valence electrons in the picture.
Periods in the periodic table. 6 Labeled Periodic Table Of Elements With Groups Of Groups. Groups and periods are two most important methods of classifying the elements in the periodic table.
The most common way the periodic table is classified by metals nonmetals and metalloids. This is what is meant by periodicity or periodic table trends. For example all the elements of group 2 have an electron configuration of E ns 2 where E is an inert gas configuration and have notable similarities in their chemical properties.
Copy the picture below 2.
:max_bytes(150000):strip_icc()/PeriodicTableCrystal-56a12d9b5f9b58b7d0bccfdf.png)
Why Is The Periodic Table Color Coded
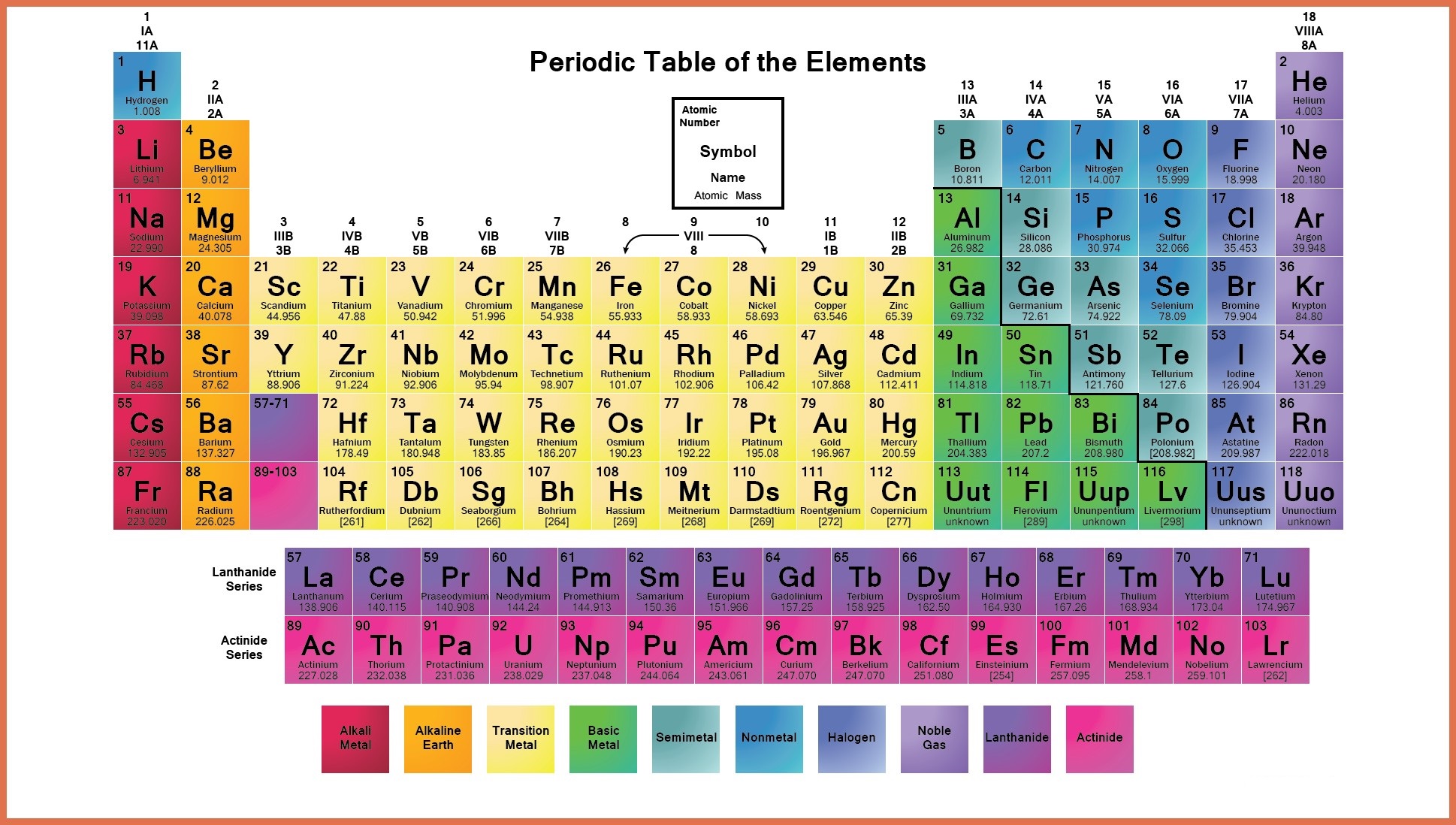
Labeled Periodic Table Of Elements With Name
Ks3 Ks4 Periodic Table Colour Coded Groups Periods Teaching Resources

The Periodic Table Properties Of Groups And Periods Science Class Video Study Com
How To Identify Groups Periods And Blocks Of Elements On The Basis Of Electronic Configuration Quora